|
8/27/2023 0 Comments Explain charles law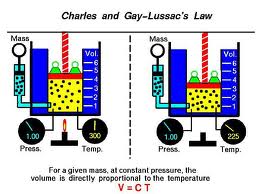
The ideal gas equation (1) P V n R T describes how gases behave, e.g. The physical principle known as Charles' law states that the volume of a gas equals a constant value multiplied by its temperature as measured on the Kelvin scale (zero Kelvin corresponds to. To use Kinetic Molecular Theory to describe the behavior of the macroscopic gas laws. V 1 = 750 mm 3, T 1 = 55 ☌, V 2 = ?, T 2 = 21 ☌ Basics of Kinetic Molecular Theory Common Ideal Gas Difficulties Learning Objectives To understand the five fundamentals of Kinetic Molecular Theory.The data is as follows: Volume vs temperature experimental data Volume (mL)įind whether the gas obeys Charles' law by plotting the graph of volume vs temperature? If yes, determine absolute zero? Problem 11įind the missing quantity from the following data. The volume-temperature data for a fixed amount of the gas is recorded. What is the volume in the icy water? Problem 10Īn experiment is performed on a gas under constant pressure to studies its behaviour. The volume of the air in the syringe at room temperature (20 ☌) is 53 mL. What will be the temperature at 700 mL if the pressure is constant? Problem 9Ī sealed syringe dipped in icy water at a temperature of −12 ☌. Find the new volume if the temperature is reduced to 220 K? Problem 8Ī student collects an experimental gas of a volume of 730 mL at 84 ☌. Los Angles to Death Valley Problem 7Ī gas occupies 22.2 mL at a temperature of 358 K. Find the volume of the balloon in Death Valley where the temperature is 51 ☌? The pressure is constant at both places. Discover how this law explains the behavior of gases and how they expand or contract in response to. The volume of a balloon of helium is 0.75 L in Los Angles. Learn about Charles law, named for Jacques Charles who introduced the law circa 1780. If the volume of the gas is double, what will be its new temperature? Problem 6 The volume of oxygen gas at 25 ☌ is 10 m 3. Find the final volume if the initial volume is 4.0 m 3? Problem 5 Find the initial volume? Problem 4ĭuring transport of steam from one industrial vessel to another, temperature drops by 5 ☌. The temperature of a gas changes from 25 ☌ to 80 ☌. Determine the initial temperature if the expansion is isobaric? Problem 3 Find the final temperature of the gas? Problem 2Ĭarbon dioxide gas expands from 10.0 cm 3 to 11.5 cm 3. Hydrogen gas contracts at constant pressure from 1.00 L to 0.95 L. How Charles' law and absolute zero are related? Problems What happens to the temperature of the gas? Question 7 
The volume of a fixed amount of gas is double under constant pressure. Give some real-life examples of Charles' law? Question 4Įxplain graphs of Charles' law? Question 5Ĭan Charles' law be experimentally proven? If yes, write in a few words how would you? Question 6 Is Charles' law universally true? If not, what are its limitations? Question 3 Question 1Įxplain Charles' law with its equation? Question 2 Analysis of a gas when its temperature increases according to kinetic theory: The temperature has increased therefore the molecules have more kinetic energy, so they move with a greater velocity. If you think it is necessary, you may use illustrations to support your answer. Charles' law: for a fixed mass of gas at constant pressure the volume is directly proportional to the temperature. The average kinetic energy of the particles in a gas is directly proportional to the Kelvin temperature of the gas.Write a brief answer to the following questions. > Explanation of Charle’s law - This law means that with the increase in. > Statement of Charle’s law - According to this law, at constant pressure and for constant mass of a gas, the volume of a gas is directly proportional to the temperature. This temperature is known as absolute zero. Complete step by step answer: First we will state Charles law and then we will explain it. Įventually at absolute zero, which is -273☌, the molecules stop moving and the gas does not exert any pressure at all. This means that the pressure also decreases. When the temperature is decreased, the gas particles move more slowly and the collisions are less hard and less frequent.This means that the pressure also increases. When the temperature is increased, the gas particles move faster and the collisions become harder and more frequent.What happens if we change the temperature of a fixed volume of gas? So, increasing pressure means that volume will decrease providing that the temperature remains constant. Boyle’s Lawīoyle’s Law states that for a gas at a constant temperature, pressure × volume is also constant. So, for a fixed mass of gas at a constant pressure, volume ÷ temperature will remain the same. Charles’ Law states that if a gas is heated up and the pressure does not change, the volume will.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
